
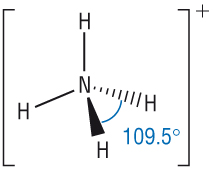
These bonds are slightly different from that in a hydrogen molecule. Most chemical bonds are not between like atoms but form between atoms of different elements. Covalent, Polar covalent, and Ionic Bondsīecause the hydrogen molecule contains two identical atoms, it can be assumed that the bonding electrons in this covalent bond are shared equally by these atoms. This release of energy shows that the molecule of hydrogen is more stable than the separate atoms.Ī. When this bond forms, energy is released. This chemical bond is a covalent bond,Ī pair of electrons shared between two atoms. These shared electrons form a bond between the two atoms. In the resulting molecule, both atoms have two electrons and a filled outer (valence) shell. Although the electrons may be in any part of this orbital, we can predict that they are most likely to be in the space between the nuclei, shielding one nucleus from the other and being attracted by both. This orbital encompasses space around both nuclei. Eventually the two orbitals overlap, becoming a single orbital containing two electrons (see Figure 7.1).įIGURE 7.1 Two hydrogen atoms, each with one electron, combine to form a hydrogen molecule, in which the two electrons are shared between the atoms and serve to give each atom a filled valence shell. As the two atoms approach each other, the nucleus of one atom attracts the electron of the other. The simplest chemical bond is that formed between two hydrogen atoms. Thus the outer shell of each bonded atom will contain eight electrons (or two electrons for hydrogen and lithium). According to the octet rule Section 5.7C1,Ītoms bond together to form molecules in such a way that each atom participating in a chemical bond acquires an electron configuration resembling that of the noble gas nearest it in the periodic table. Another compound with similar properties is ammonium polysulfide.The atoms of a compound are held together byįormed by the interaction of electrons from each atom. It is highly flammable and should be stored away from fire or other flammable liquids and substances.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
